Lacnotuzumab: Unveiling the Role of Anti-CD47 in Cancer Research
Quick Facts About Lacnotuzumab
What is Lacnotuzumab?
What is the mechanism of action for Lacnotuzumab?
By blocking CD47, Lacnotuzumab enhances macrophage-mediated phagocytosis, allowing the immune system to recognize and eliminate cancer cells.
What are the clinical applications of Lacnotuzumab?
Lacnotuzumab has been studied in clinical trials for its potential in treating solid tumors and hematologic malignancies, often in combination with other immunotherapies.
1.) Understanding Lacnotuzumab
Lacnotuzumab is an emerging therapeutic agent within the field of immuno-oncology, specifically designed to target CD47, a protein that plays a critical role in immune evasion by tumor cells. CD47 acts as a “don’t eat me” signal, inhibiting macrophages from recognizing and attacking cancer cells. Many tumors overexpress CD47 to avoid immune surveillance, allowing them to grow uncontrollably. Lacnotuzumab works by blocking the interaction between CD47 and its receptor, SIRPα, found on macrophages. This disruption restores immune activity, enabling macrophages to identify, ingest, and destroy tumor cells, thereby reactivating the body’s natural defense mechanisms.
What sets Lacnotuzumab apart from traditional cancer therapies is its ability to modulate the immune system rather than directly targeting cancer cells. Conventional chemotherapy treatments typically affect both malignant and healthy cells, resulting in significant side effects. Lacnotuzumab, on the other hand, works by leveraging the body’s immune response to selectively target and eliminate cancer cells, potentially reducing the systemic toxicity often seen with conventional chemotherapies. This immune-based approach represents a promising avenue for treating cancers that have become resistant to traditional therapies, particularly those with high levels of CD47 expression.
The growing interest in immune-modulatory cancer treatments underscores the potential of Lacnotuzumab as a key player in immunotherapy. Ongoing studies are focused on optimizing its effectiveness, exploring its role in combination with other immune-based therapies, and identifying patient populations that would benefit the most. As research advances, Lacnotuzumab may offer a transformative approach to cancer treatment, particularly for hard-to-treat cancers.
2.) Mechanism of Action of Lacnotuzumab
Lacnotuzumab functions by specifically binding to CD47, a protein expressed on the surface of many cancer cells. CD47 interacts with the SIRPα receptor on macrophages, sending a signal that prevents these immune cells from phagocytosing (engulfing and destroying) the cancer cells. This interaction is a critical mechanism by which tumors evade immune detection, allowing them to proliferate unchecked. Lacnotuzumab intervenes in this immune evasion process by blocking the interaction between CD47 and SIRPα, preventing the immune suppression that tumors rely on to avoid immune clearance.
By inhibiting this “don’t eat me” signal, Lacnotuzumab reactivates the immune system's ability to recognize, target, and destroy cancer cells. The drug's action is particularly beneficial in cancers where CD47 is overexpressed, such as leukemia, lymphoma, and certain solid tumors. This immune-mediated mechanism is distinct from that of many traditional cancer therapies, which primarily target cancer cells directly with cytotoxic agents, often leading to collateral damage to healthy tissues. Instead, Lacnotuzumab works by enhancing the body’s natural immune response, which can provide more targeted and, in some cases, more efficient treatment.
Additionally, Lacnotuzumab’s ability to modify the immune environment opens up possibilities for combination therapy with other immune-based treatments, such as immune checkpoint inhibitors. This combination could potentially enhance its effectiveness, especially in cancers that are resistant to standard therapies. As research into Lacnotuzumab continues, the goal is to refine its dosage and identify the optimal patient populations that would benefit the most from this innovative immune-modulatory approach.
3.) Clinical Applications of Lacnotuzumab
Lacnotuzumab is being evaluated in clinical trials as part of a broader effort to explore the potential of anti-CD47 therapies in oncology. The drug has been tested in various settings, with particular focus on hematologic malignancies such as leukemia and lymphoma, as well as certain solid tumors. In these cancers, CD47 is often overexpressed, allowing tumor cells to evade immune detection and destruction. By inhibiting CD47, Lacnotuzumab restores immune surveillance, offering a novel strategy to combat these cancers.
Early-phase trials have provided important insights into Lacnotuzumab’s pharmacokinetics and potential therapeutic efficacy. However, some trials have been discontinued due to challenges such as safety concerns or insufficient patient response. Despite these setbacks, the research continues to progress, with newer studies focusing on optimizing dosing strategies, refining treatment regimens, and exploring combination therapies. Lacnotuzumab’s role in personalized medicine is also being investigated, as biomarkers could help identify patients most likely to benefit from CD47 blockade. For instance, determining the level of CD47 expression in tumors may provide a predictive tool for treatment efficacy, making Lacnotuzumab a part of a broader personalized cancer therapy approach.
Moreover, ongoing studies are examining the potential of Lacnotuzumab in combination with other immunotherapeutic agents, such as immune checkpoint inhibitors. Combining Lacnotuzumab with drugs like PD-1 or PD-L1 inhibitors could enhance the immune system’s ability to target cancer cells, especially in cancers that have developed resistance to standard therapies. As the field of immuno-oncology evolves, Lacnotuzumab holds promise for improving outcomes in patients with difficult-to-treat cancers, and future clinical trials will continue to refine its role in cancer treatment.
4.) Exploring Biosimilars for Lacnotuzumab
What is a Biosimilar?
A biosimilar is a biologic product highly similar to an already approved reference biologic, with no clinically meaningful differences in safety, efficacy, or purity. Biosimilars provide cost-effective alternatives for research and clinical applications, enabling broader access to advanced therapeutics.
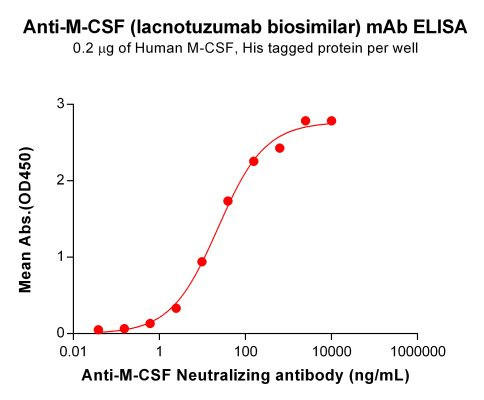
| Lacnotuzumab (Anti-M-CSF) Biosimilar Antibody | |
|---|---|
| Antibody Type: | Monoclonal Antibody |
| Protein: | M-CSF |
| Reactivity: | Human |
How Lacnotuzumab Biosimilar Compares to Lacnotuzumab
Biosimilars of Lacnotuzumab offer comparable binding affinity, functional activity, and safety profiles. While the reference product has been the focus of clinical trials, biosimilars serve as essential tools for preclinical research, allowing scientists to investigate novel applications and combinations.
Benefits of Lacnotuzumab Biosimilars in Research
- Cost-Effective Research Tools: Biosimilars reduce financial barriers for institutions studying anti-CD47 therapies.
- Consistency and Availability: They provide a reliable supply for research, ensuring experimental reproducibility.
- Expanding Scientific Discoveries: Researchers can explore combination therapies, resistance mechanisms, and biomarker-driven treatment strategies.
Research Use Only Disclaimer:
Lacnotuzumab biosimilar is intended for research purposes only and is not approved for clinical use in patients. Researchers should ensure compliance with regulatory guidelines when utilizing biosimilars in experimental studies.
Discover Our Biosimilar Range
At Assay Genie, we specialize in providing high-quality biosimilars for research use! Check out our full biosimilar range to learn more.

By David Lee, PhD
Recent Posts
-
Enavatuzumab: Revolutionizing Cancer Research Through Novel Therapeutics
Quick Facts About EnavatuzumabWhat is Enavatuzumab?Enavatuzumab is a monoclonal antibo …17th Dec 2025 -
Alemtuzumab: Mechanism, Applications, and Biosimilar Advancements
Quick Facts About AlemtuzumabWhat is Alemtuzumab?Alemtuzumab is a monoclonal antibody …17th Dec 2025 -
Erenumab: Transforming Migraine Prevention Through CGRP Receptor Inhibition
Quick Facts About ErenumabWhat is Erenumab?Erenumab is a fully human monoclonal antibo …1st Apr 2025


