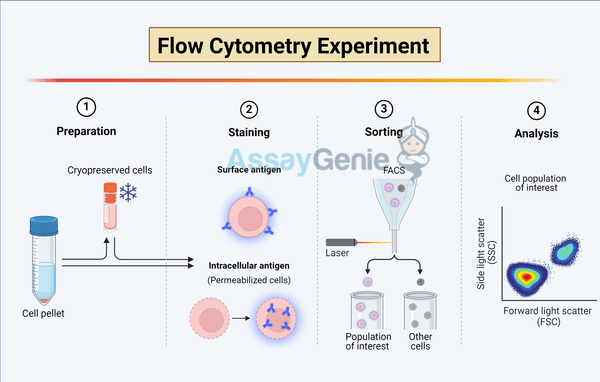
Navigating the Intricacies of Intracellular Flow Cytometry: Key Considerations
In the evolving landscape of cellular biology, intracellular flow cytometry (ICFC) stands out as a powerful technique for analyzing the complexities within cells. This advanced method extends the capabilities of traditional flow cytometry by allowing researchers to investigate intracellular proteins, signaling pathways, and specific gene expressions within individual cells. ICFC has become indispensable in various fields, including immunology, oncology, and drug discovery, offering insights that are pivotal for understanding disease mechanisms and developing therapeutic strategies. However, harnessing the full potential of ICFC requires meticulous attention to several critical considerations. This article delves into three primary aspects that researchers must navigate to optimize ICFC applications: cell permeabilization, antibody specificity and titration, and compensation strategies for fluorescence overlap.
1. Cell Permeabilization: A Gateway to the Intracellular Environment
A fundamental step in ICFC is permeabilizing the cell membrane to allow antibodies to access intracellular targets. This process is delicate; it requires a balance between maintaining cell integrity and permitting entry of detection reagents. The choice of permeabilization method depends on the nature of the intracellular molecules of interest and the cell type. Commonly used agents include saponin, which is reversible and maintains cellular morphology by creating pores in the membrane, and methanol or formaldehyde, which fix and permeabilize cells by cross-linking proteins but may alter or mask epitopes.
Considerations:
- Epitope Preservation: Selecting a permeabilization technique that preserves the integrity of the target epitopes is crucial for accurate detection.
- Cell Viability: For live cell analysis, viability dyes that exclude dead cells can be used to ensure that the data reflect the behavior of live cells only.
2. Antibody Specificity and Titration: Ensuring Precision in Detection
The specificity and proper titration of antibodies are paramount in ICFC to achieve accurate and reproducible results. Antibodies that bind specifically to the target antigen without cross-reacting with other proteins ensure that the signals detected correspond to the intended intracellular markers. Furthermore, titration is essential to determine the optimal concentration of antibody that provides the maximum signal-to-noise ratio, thereby enhancing the sensitivity and specificity of detection.
Considerations:
- Fluorochrome Selection: Choosing antibodies conjugated with fluorochromes that match the detectors available on the flow cytometer and minimize spectral overlap is crucial.
- Validation: It is imperative to validate the specificity of antibodies, particularly when studying proteins with close homologs or in complex intracellular pathways.
3. Compensation Strategies for Fluorescence Overlap: Mastering the Art of Discrimination
Fluorescence compensation is a critical aspect of ICFC, especially when multiple fluorochromes are used. Spectral overlap occurs when the emission spectrum of one fluorochrome spills into the detector of another, potentially confounding analysis. Proper compensation allows for the accurate discrimination of signals from different fluorochromes, ensuring that each signal is correctly attributed to its respective fluorochrome.
Considerations:
- Compensation Controls: Using single-stained controls for each fluorochrome is essential for setting accurate compensation settings.
- Software Tools: Leveraging advanced flow cytometry software that can automate and refine compensation based on control samples can significantly improve data accuracy and analysis efficiency.
Beyond the Basics: Advanced Considerations in ICFC
While the aforementioned considerations are foundational, advancing ICFC techniques further involves delving into more complex aspects, such as:
- Intracellular Cytokine Staining: Detecting cytokines within cells requires careful fixation and permeabilization to preserve these transiently expressed proteins without causing them to leak out of the cell.
- Phospho-Protein Analysis: Investigating phosphorylation states of proteins involved in signaling pathways necessitates the use of phospho-specific antibodies and conditions that prevent dephosphorylation post-cell fixation.
Conclusion
References
- Shapiro, H.M. (2018). Practical Flow Cytometry, 5th Edition. Wiley-Liss.
- Perfetto, S.P., Chattopadhyay, P.K., & Roederer, M. (2004). Seventeen-colour flow cytometry: unravelling the immune system. Nature Reviews Immunology, 4(8), 648-655.
- Herzenberg, L.A., Parks, D., Sahaf, B., Perez, O., Roederer, M., & Herzenberg, L.A. (2002). The history and future of the Fluorescence Activated Cell Sorter and Flow Cytometry: A view from Stanford. Clinical Chemistry, 48(10), 1819-1827.
- Krutzik, P.O., & Nolan, G.P. (2003). Intracellular phospho-protein staining techniques for flow cytometry: monitoring single cell signaling events. Cytometry Part A, 55(2), 61-70.
- Saeys, Y., Van Gassen, S., & Lambrecht, B.N. (2016). Computational flow cytometry: helping to make sense of high-dimensional immunology data. Nature Reviews Immunology, 16(7), 449-462.
- Maecker, H.T., McCoy, J.P., & Nussenblatt, R. (2012). Standardizing immunophenotyping for the Human Immunology Project. Nature Reviews Immunology, 12(3), 191-200.
- Zivanovic, S., Racki, N., & Filipovic, N. (2019). Advancements in the Application of Flow Cytometry for Evaluating Intracellular Conditions. Trends in Biotechnology, 37(11), 1260-1274.
- Chattopadhyay, P.K., Hogerkorp, C.M., & Roederer, M. (2008). A chromatic explosion: the development and future of multiparameter flow cytometry. Immunology, 125(4), 441-449.
Written by Tehreem Ali
Tehreem Ali completed her MS in Bioinformatics and conducted her research work at the IOMM lab at GCUF, Pakistan.
Recent Posts
-
Enavatuzumab: Revolutionizing Cancer Research Through Novel Therapeutics
Quick Facts About EnavatuzumabWhat is Enavatuzumab?Enavatuzumab is a monoclonal antibo …17th Dec 2025 -
Alemtuzumab: Mechanism, Applications, and Biosimilar Advancements
Quick Facts About AlemtuzumabWhat is Alemtuzumab?Alemtuzumab is a monoclonal antibody …17th Dec 2025 -
Validation of MycoGenie Rapid Mycoplasma Detection Kit - A highly sensitive visual determination method for Mycoplasma detection.
The MycoGenie Rapid Mycoplasma Detection Kit enables the detection of 28 Mycoplasma sp …3rd Mar 2025