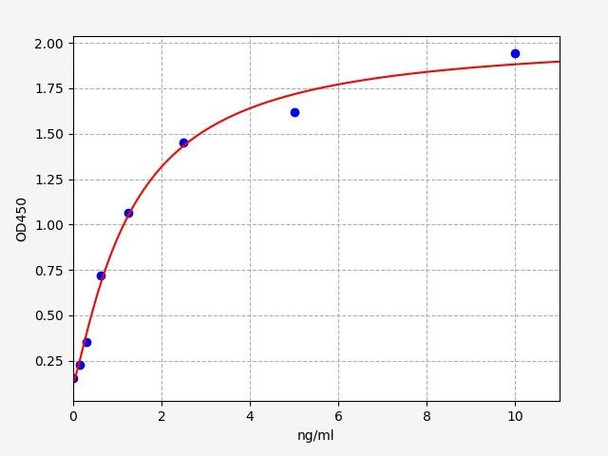
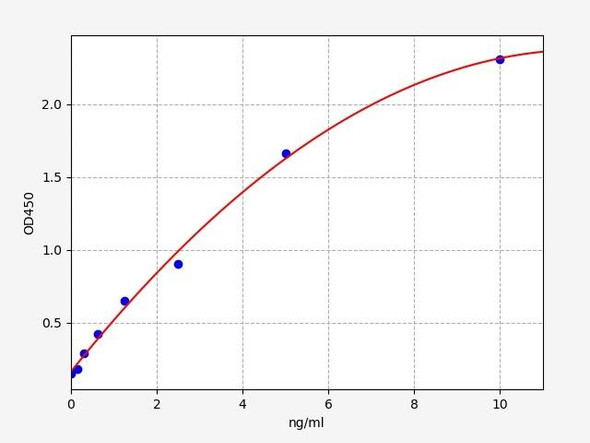
Human LMNA (Lamin-A) ELISA Kit (HUFI05107)
- SKU:
- HUFI05107
- Product Type:
- ELISA Kit
- Size:
- 96 Assays
- Uniprot:
- P02545
- Sensitivity:
- 0.094ng/ml
- Range:
- 0.156-10ng/ml
- ELISA Type:
- Sandwich
- Synonyms:
- 70 kDa lamin, CDCD1, CDDC, CMD1A, CMT2B1, EMD2, FPL, FPLD, HGPS, IDC, lamin A, lamin A, C, LDP1, LFP, LGMD1B, LMN1, LMNA, LMNC, Prelamin A, C, PRO1, progerin
- Reactivity:
- Human
Description
Human LMNA (Lamin-A) ELISA Kit
The Human LMNA (Lamin A) ELISA Kit is specifically designed to accurately measure levels of Lamin A in human serum, plasma, and cell culture supernatants. This kit guarantees high sensitivity and specificity, ensuring precise and consistent results for various research purposes.Lamin A is a key protein involved in maintaining the structural integrity of the cell nucleus and regulating gene expression. Abnormalities in Lamin A have been linked to a variety of genetic disorders known as laminopathies, including muscular dystrophy and premature aging syndromes.
By accurately quantifying Lamin A levels, researchers can gain insights into the mechanisms underlying these diseases and potentially develop targeted treatments.Overall, the Human LMNA (Lamin A) ELISA Kit is an essential tool for researchers studying nuclear envelope biology, genetic disorders, and potential therapeutic interventions. Its reliable performance and ease of use make it a valuable asset in the field of biomedical research.
| Product Name: | Human LMNA (Lamin-A) ELISA Kit |
| Product Code: | HUFI05107 |
| Size: | 96 Assays |
| Alias: | 70 kDa lamin ELISA Kit, CDCD1 ELISA Kit, CDDC ELISA Kit, CMD1A ELISA Kit, CMT2B1 ELISA Kit, EMD2 ELISA Kit, FPL ELISA Kit, FPLD ELISA Kit, HGPS ELISA Kit, IDC ELISA Kit, lamin A ELISA Kit, lamin A ELISA Kit, C ELISA Kit, LDP1 ELISA Kit, LFP ELISA Kit, LGMD1B ELISA Kit, LMN1 ELISA Kit, LMNA ELISA Kit, LMNC ELISA Kit, Prelamin A ELISA Kit, C ELISA Kit, PRO1 ELISA Kit, progerin ELISA Kit |
| Detection method: | Sandwich ELISA, Double Antibody |
| Application: | This immunoassay kit allows for the in vitro quantitative determination of Human LMNA (Lamin-A) concentrations in serum plasma and other biological fluids. |
| Sensitivity: | < 0.094ng/ml |
| Range: | 0.156-10ng/ml |
| Storage: | 4°C for 6 months |
| Note: | For Research Use Only |
| Recovery: | Matrices listed below were spiked with certain level of Human LMNA (Lamin-A) and the recovery rates were calculated by comparing the measured value to the expected amount of Human LMNA (Lamin-A) in samples. Enquire for more information. |
| Linearity: | The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Human LMNA (Lamin-A) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected. Enquire for more information. |
| CV(%): | Intra-Assay: CV<8% Inter-Assay: CV<10% |
| Component | Quantity | Storage |
| ELISA Microplate (Dismountable) | 8×12 strips | 4°C for 6 months |
| Lyophilized Standard | 2 | 4°C/-20°C |
| Sample/Standard Dilution Buffer | 20ml | 4°C |
| Biotin-labeled Antibody(Concentrated) | 120ul | 4°C (Protect from light) |
| Antibody Dilution Buffer | 10ml | 4°C |
| HRP-Streptavidin Conjugate(SABC) | 120ul | 4°C (Protect from light) |
| SABC Dilution Buffer | 10ml | 4°C |
| TMB Substrate | 10ml | 4°C (Protect from light) |
| Stop Solution | 10ml | 4°C |
| Wash Buffer(25X) | 30ml | 4°C |
| Plate Sealer | 5 | - |
Other materials and equipment required:
- Microplate reader with 450 nm wavelength filter
- Multichannel Pipette, Pipette, microcentrifuge tubes and disposable pipette tips
- Incubator
- Deionized or distilled water
- Absorbent paper
- Buffer resevoir
| UniProt Protein Function: | Function: Lamins are components of the nuclear lamina, a fibrous layer on the nucleoplasmic side of the inner nuclear membrane, which is thought to provide a framework for the nuclear envelope and may also interact with chromatin. Lamin A and C are present in equal amounts in the lamina of mammals. Plays an important role in nuclear assembly, chromatin organization, nuclear membrane and telomere dynamics. Ref.24 Ref.25Prelamin-A/C can accelerate smooth muscle cell senescence. It acts to disrupt mitosis and induce DNA damage in vascular smooth muscle cells (VSMCs), leading to mitotic failure, genomic instability, and premature senescence. Ref.24 Ref.25 |
| UniProt Protein Details: | Subunit structure: Homodimer of lamin A and lamin C. Interacts with lamin-associated polypeptides IA, IB and TMPO-alpha, RB1 and with emerin. Interacts with SREBF1, SREBF2, SUN2 and TMEM43 By similarity. Proteolytically processed isoform A interacts with NARF. Interacts with SUN1. Prelamin-A/C interacts with EMD. Interacts with MLIP; may regulate MLIP localization to the nucleus envelope. Interacts with DMPK; may regulate nuclear envelope stability. Ref.12 Ref.13 Ref.22 Ref.26 Ref.29 Ref.30 Subcellular location: Nucleus. Nucleus envelope. Note: Farnesylation of prelamin-A/C facilitates nuclear envelope targeting and subsequent cleaveage by ZMPSTE24/FACE1 to remove the farnesyl group produces mature lamin-A/C, which can then be inserted into the nuclear lamina. EMD is required for proper localization of non-farnesylated prelamin-A/C. Ref.17 Ref.22 Tissue specificity: In the arteries, prelamin-A/C accumulation is not observed in young healthy vessels but is prevalent in medial vascular smooth muscle cells (VSMCs) from aged individuals and in atherosclerotic lesions, where it often colocalizes with senescent and degenerate VSMCs. Prelamin-A/C expression increases with age and disease. In normal aging, the accumulation of prelamin-A/C is caused in part by the down-regulation of ZMPSTE24/FACE1 in response to oxidative stress. Ref.25 Post-translational modification: Increased phosphorylation of the lamins occurs before envelope disintegration and probably plays a role in regulating lamin associations.Proteolytic cleavage of the C-terminal of 18 residues of prelamin-A/C results in the production of lamin-A/C. The prelamin-A/C maturation pathway includes farnesylation of CAAX motif, ZMPSTE24/FACE1 mediated cleavage of the last three amino acids, methylation of the C-terminal cysteine and endoproteolytic removal of the last 15 C-terminal amino acids. Proteolytic cleavage requires prior farnesylation and methylation, and absence of these blocks cleavage.Sumoylation is necessary for the localization to the nuclear envelope. Ref.17Farnesylation of prelamin-A/C facilitates nuclear envelope targeting. Involvement inDisease: Emery-Dreifuss muscular dystrophy 2, autosomal dominant (EDMD2) [MIM:181350]: A form of Emery-Dreifuss muscular dystrophy, a degenerative myopathy characterized by weakness and atrophy of muscle without involvement of the nervous system, early contractures of the elbows, Achilles tendons and spine, and cardiomyopathy associated with cardiac conduction defects.Note: The disease is caused by mutations affecting the gene represented in this entry. Ref.26 Ref.35 Ref.39 Ref.40 Ref.44 Ref.45 Ref.47 Ref.55 Ref.64 Ref.67 Ref.73 Ref.77 Ref.87Emery-Dreifuss muscular dystrophy 3, autosomal recessive (EDMD3) [MIM:181350]: A form of Emery-Dreifuss muscular dystrophy, a degenerative myopathy characterized by weakness and atrophy of muscle without involvement of the nervous system, early contractures of the elbows, Achilles tendons and spine, and cardiomyopathy associated with cardiac conduction defects.Note: The disease is caused by mutations affecting the gene represented in this entry. Ref.88Cardiomyopathy, dilated 1A (CMD1A) [MIM:115200]: A disorder characterized by ventricular dilation and impaired systolic function, resulting in congestive heart failure and arrhythmia. Patients are at risk of premature death.Note: The disease is caused by mutations affecting the gene represented in this entry. Ref.17 Ref.36 Ref.46 Ref.47 Ref.49 Ref.54 Ref.59 Ref.63 Ref.64 Ref.68 Ref.69 Ref.78 Ref.86Lipodystrophy, familial partial, 2 (FPLD2) [MIM:151660]: A disorder characterized by the loss of subcutaneous adipose tissue in the lower parts of the body (limbs, buttocks, trunk). It is accompanied by an accumulation of adipose tissue in the face and neck causing a double chin, fat neck, or cushingoid appearance. Adipose tissue may also accumulate in the axillae, back, labia majora, and intraabdominal region. Affected patients are insulin-resistant and may develop glucose intolerance and diabetes mellitus after age 20 years, hypertriglyceridemia, and low levels of high density lipoprotein cholesterol.Note: The disease is caused by mutations affecting the gene represented in this entry. Ref.37 Ref.41 Ref.43 Ref.47 Ref.53 Ref.56 Ref.61 Ref.81Limb-girdle muscular dystrophy 1B (LGMD1B) [MIM:159001]: An autosomal dominant degenerative myopathy with age-related atrioventricular cardiac conduction disturbances, dilated cardiomyopathy, and the absence of early contractures. Characterized by slowly progressive skeletal muscle weakness of the hip and shoulder girdles. Muscle biopsy shows mild dystrophic changes.Note: The disease is caused by mutations affecting the gene represented in this entry. Ref.42 Ref.48 Ref.55 Ref.58 Ref.77 Ref.83Charcot-Marie-Tooth disease 2B1 (CMT2B1) [MIM:605588]: A recessive axonal form of Charcot-Marie-Tooth disease, a disorder of the peripheral nervous system, characterized by progressive weakness and atrophy, initially of the peroneal muscles and later of the distal muscles of the arms. Charcot-Marie-Tooth disease is classified in two main groups on the basis of electrophysiologic properties and histopathology: primary peripheral demyelinating neuropathies (designated CMT1 when they are dominantly inherited) and primary peripheral axonal neuropathies (CMT2). Neuropathies of the CMT2 group are characterized by signs of axonal regeneration in the absence of obvious myelin alterations, normal or slightly reduced nerve conduction velocities, and progressive distal muscle weakness and atrophy. Nerve conduction velocities are normal or slightly reduced.Note: The disease is caused by mutations affecting the gene represented in this entry. Ref.50Hutchinson-Gilford progeria syndrome (HGPS) [MIM:176670]: Rare genetic disorder characterized by features reminiscent of marked premature aging.Note: The disease is caused by mutations affecting the gene represented in this entry. HGPS is caused by the toxic accumulation of a mutant form of lamin-A/C. This mutant protein, called progerin, acts to deregulate mitosis and DNA damage signaling, leading to premature cell death and senescence. Progerin lacks the conserved ZMPSTE24/FACE1 cleavage site and therefore remains permanently farnesylated. Thus, although it can enter the nucleus and associate with the nuclear envelope, it cannot incorporate normally into the nuclear lamina. Ref.26 Ref.62 Ref.65 Ref.66 Ref.72 Ref.74Cardiomyopathy, dilated, with hypergonadotropic hypogonadism (CMDHH) [MIM:212112]: A disorder characterized by the association of genital anomalies, hypergonadotropic hypogonadism and dilated cardiomyopathy. Patients can present other variable clinical manifestations including mental retardation, skeletal anomalies, scleroderma-like skin, graying and thinning of hair, osteoporosis. Dilated cardiomyopathy is characterized by ventricular dilation and impaired systolic function, resulting in congestive heart failure and arrhythmia.Note: The disease is caused by mutations affecting the gene represented in this entry. Ref.65 Ref.80 Ref.85Mandibuloacral dysplasia with type A lipodystrophy (MADA) [MIM:248370]: A disorder characterized by mandibular and clavicular hypoplasia, acroosteolysis, delayed closure of the cranial suture, progeroid appearance, partial alopecia, soft tissue calcinosis, joint contractures, and partial lipodystrophy with loss of subcutaneous fat from the extremities. Adipose tissue in the face, neck and trunk is normal or increased.Note: The disease is caused by mutations affecting the gene represented in this entry. Ref.52 Ref.76 Ref.79Lethal tight skin contracture syndrome (LTSCS) [MIM:275210]: Rare disorder mainly characterized by intrauterine growth retardation, tight and rigid skin with erosions, prominent superficial vasculature and epidermal hyperkeratosis, facial features (small mouth, small pinched nose and micrognathia), sparse/absent eyelashes and eyebrows, mineralization defects of the skull, thin dysplastic clavicles, pulmonary hypoplasia, multiple joint contractures and an early neonatal lethal course. Liveborn children usually die within the first week of life. The overall prevalence of consanguineous cases suggested an autosomal recessive inheritance.Note: The disease is caused by mutations affecting the gene represented in this entry. Ref.70Heart-hand syndrome Slovenian type (HHS-Slovenian) [MIM:610140]: Heart-hand syndrome (HHS) is a clinically and genetically heterogeneous disorder characterized by the co-occurrence of a congenital cardiac disease and limb malformations.Note: The disease is caused by mutations affecting the gene represented in this entry.Muscular dystrophy congenital LMNA-related (MDCL) [MIM:613205]: A form of congenital muscular dystrophy. Patients present at birth, or within the first few months of life, with hypotonia, muscle weakness and often with joint contractures.Note: The disease is caused by mutations affecting the gene represented in this entry. Ref.84 Miscellaneous: There are three types of lamins in human cells: A, B, and C.The structural integrity of the lamina is strictly controlled by the cell cycle, as seen by the disintegration and formation of the nuclear envelope in prophase and telophase, respectively. Sequence similarities: Belongs to the intermediate filament family. Sequence caution: The sequence CAA27173.1 differs from that shown. Reason: Frameshift at position 582. |
| NCBI Summary: | The nuclear lamina consists of a two-dimensional matrix of proteins located next to the inner nuclear membrane. The lamin family of proteins make up the matrix and are highly conserved in evolution. During mitosis, the lamina matrix is reversibly disassembled as the lamin proteins are phosphorylated. Lamin proteins are thought to be involved in nuclear stability, chromatin structure and gene expression. Vertebrate lamins consist of two types, A and B. Alternative splicing results in multiple transcript variants. Mutations in this gene lead to several diseases: Emery-Dreifuss muscular dystrophy, familial partial lipodystrophy, limb girdle muscular dystrophy, dilated cardiomyopathy, Charcot-Marie-Tooth disease, and Hutchinson-Gilford progeria syndrome. [provided by RefSeq, Apr 2012] |
| UniProt Code: | P02545 |
| NCBI GenInfo Identifier: | 125962 |
| NCBI Gene ID: | 4000 |
| NCBI Accession: | P02545.1 |
| UniProt Secondary Accession: | P02545,P02546, Q5TCJ2, Q5TCJ3, Q969I8, Q96JA2, B4DI32 D3DVB0, E7EUI9, |
| UniProt Related Accession: | P02545 |
| Molecular Weight: | 74,139 Da |
| NCBI Full Name: | Prelamin-A/C |
| NCBI Synonym Full Names: | lamin A/C |
| NCBI Official Symbol: | LMNA |
| NCBI Official Synonym Symbols: | FPL; IDC; LFP; CDDC; EMD2; FPLD; HGPS; LDP1; LMN1; LMNC; PRO1; CDCD1; CMD1A; FPLD2; LMNL1; CMT2B1; LGMD1B |
| NCBI Protein Information: | lamin; 70 kDa lamin; prelamin-A/C; lamin A/C-like 1; renal carcinoma antigen NY-REN-32 |
| UniProt Protein Name: | Prelamin-A/C |
| UniProt Synonym Protein Names: | 70 kDa lamin; Renal carcinoma antigen NY-REN-32 |
| Protein Family: | Prelamin |
| UniProt Gene Name: | LMNA |
| UniProt Entry Name: | LMNA_HUMAN |
*Note: Protocols are specific to each batch/lot. For the correct instructions please follow the protocol included in your kit.
Before adding to wells, equilibrate the SABC working solution and TMB substrate for at least 30 min at 37 °C. When diluting samples and reagents, they must be mixed completely and evenly. It is recommended to plot a standard curve for each test.
| Step | Protocol |
| 1. | Set standard, test sample and control (zero) wells on the pre-coated plate respectively, and then, record their positions. It is recommended to measure each standard and sample in duplicate. Wash plate 2 times before adding standard, sample and control (zero) wells! |
| 2. | Aliquot 0.1ml standard solutions into the standard wells. |
| 3. | Add 0.1 ml of Sample / Standard dilution buffer into the control (zero) well. |
| 4. | Add 0.1 ml of properly diluted sample ( Human serum, plasma, tissue homogenates and other biological fluids.) into test sample wells. |
| 5. | Seal the plate with a cover and incubate at 37 °C for 90 min. |
| 6. | Remove the cover and discard the plate content, clap the plate on the absorbent filter papers or other absorbent material. Do NOT let the wells completely dry at any time. Wash plate X2. |
| 7. | Add 0.1 ml of Biotin- detection antibody working solution into the above wells (standard, test sample & zero wells). Add the solution at the bottom of each well without touching the side wall. |
| 8. | Seal the plate with a cover and incubate at 37 °C for 60 min. |
| 9. | Remove the cover, and wash plate 3 times with Wash buffer. Let wash buffer rest in wells for 1 min between each wash. |
| 10. | Add 0.1 ml of SABC working solution into each well, cover the plate and incubate at 37 °C for 30 min. |
| 11. | Remove the cover and wash plate 5 times with Wash buffer, and each time let the wash buffer stay in the wells for 1-2 min. |
| 12. | Add 90 µL of TMB substrate into each well, cover the plate and incubate at 37 °C in dark within 10-20 min. (Note: This incubation time is for reference use only, the optimal time should be determined by end user.) And the shades of blue can be seen in the first 3-4 wells (with most concentrated standard solutions), the other wells show no obvious color. |
| 13. | Add 50 µL of Stop solution into each well and mix thoroughly. The color changes into yellow immediately. |
| 14. | Read the O.D. absorbance at 450 nm in a microplate reader immediately after adding the stop solution. |
When carrying out an ELISA assay it is important to prepare your samples in order to achieve the best possible results. Below we have a list of procedures for the preparation of samples for different sample types.
| Sample Type | Protocol |
| Serum | If using serum separator tubes, allow samples to clot for 30 minutes at room temperature. Centrifuge for 10 minutes at 1,000x g. Collect the serum fraction and assay promptly or aliquot and store the samples at -80°C. Avoid multiple freeze-thaw cycles. If serum separator tubes are not being used, allow samples to clot overnight at 2-8°C. Centrifuge for 10 minutes at 1,000x g. Remove serum and assay promptly or aliquot and store the samples at -80°C. Avoid multiple freeze-thaw cycles. |
| Plasma | Collect plasma using EDTA or heparin as an anticoagulant. Centrifuge samples at 4°C for 15 mins at 1000 × g within 30 mins of collection. Collect the plasma fraction and assay promptly or aliquot and store the samples at -80°C. Avoid multiple freeze-thaw cycles. Note: Over haemolysed samples are not suitable for use with this kit. |
| Urine & Cerebrospinal Fluid | Collect the urine (mid-stream) in a sterile container, centrifuge for 20 mins at 2000-3000 rpm. Remove supernatant and assay immediately. If any precipitation is detected, repeat the centrifugation step. A similar protocol can be used for cerebrospinal fluid. |
| Cell culture supernatant | Collect the cell culture media by pipette, followed by centrifugation at 4°C for 20 mins at 1500 rpm. Collect the clear supernatant and assay immediately. |
| Cell lysates | Solubilize cells in lysis buffer and allow to sit on ice for 30 minutes. Centrifuge tubes at 14,000 x g for 5 minutes to remove insoluble material. Aliquot the supernatant into a new tube and discard the remaining whole cell extract. Quantify total protein concentration using a total protein assay. Assay immediately or aliquot and store at ≤ -20 °C. |
| Tissue homogenates | The preparation of tissue homogenates will vary depending upon tissue type. Rinse tissue with 1X PBS to remove excess blood & homogenize in 20ml of 1X PBS (including protease inhibitors) and store overnight at ≤ -20°C. Two freeze-thaw cycles are required to break the cell membranes. To further disrupt the cell membranes you can sonicate the samples. Centrifuge homogenates for 5 mins at 5000xg. Remove the supernatant and assay immediately or aliquot and store at -20°C or -80°C. |
| Tissue lysates | Rinse tissue with PBS, cut into 1-2 mm pieces, and homogenize with a tissue homogenizer in PBS. Add an equal volume of RIPA buffer containing protease inhibitors and lyse tissues at room temperature for 30 minutes with gentle agitation. Centrifuge to remove debris. Quantify total protein concentration using a total protein assay. Assay immediately or aliquot and store at ≤ -20 °C. |
| Breast Milk | Collect milk samples and centrifuge at 10,000 x g for 60 min at 4°C. Aliquot the supernatant and assay. For long term use, store samples at -80°C. Minimize freeze/thaw cycles. |