Enzyme-linked immunosorbent assay for the quantitative determination of antibodies to adalimumab in serum and plasma. Adalimumab (Humira®) was associated to the development of anti-Adalimumab antibodies, with some reported to be neutralizing in patients. This might lead to severe complications. The Assay Genie Antibody to Adalimumab ELISA Kit can be efficiently used for monitoring Adalimumab anti-drug antibodies (ADA) in biological samples and is for research use only.
Description
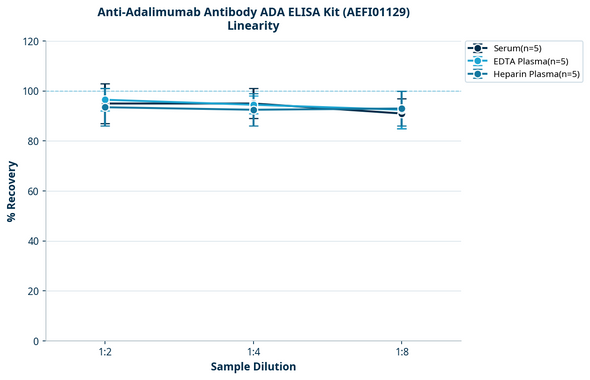
Anti-Adalimumab ADA Quantitative ELISA Kit
Anti-Adalimumab (Humira®) ADA Quantitative ELISA Kit test principle
Solid phase enzyme-linked immunosorbent assay (ELISA) based on the sandwich principle. Standards and samples (serum or plasma) are incubated in the microtiter plate coated with the reactant for adalimumab. After incubation, the wells are washed. Then, horse radish peroxidase (HRP) conjugated probe is added and binds to adalimumab captured by the reactant on the surface of the wells. Following incubation wells are washed and the bound enzymatic activity is detected by addition of tetramethylbenzidine (TMB) chromogen substrate. Finally, the reaction is terminated with an acidic stop solution. The colour developed is proportional to the amount of adalimumab in the sample or standard. Results of samples can be determined directly using the standard curve.
Anti-Adalimumab (Humira®) ADA Quantitative Product Information
| Information | Description |
Application | Free drug |
Required Volume (uL) | 10 |
Total Time (min) | 140 |
Sample Type | Serum, Plasma |
Number of Assays | 96 |
Detection Limit (ng/mL) | 30 (ng/mL) |
Spike Recovery (%) | 85-115% |
Shelf Life (year) | 1 |
Alternative Names | Tumour Necrosis Factor Alpha Humira |
Anti-Adalimumab ADA ELISA Kit
Adalimumab mode of action
Adalimumab (Humira®) is a recombinant human IgG1 monoclonal antibody specific for human tumor necrosis factor alpha (TNF-a). Adalimumab (Humira®) was created using phage display technology resulting in an antibody with human derived heavy and light chain variable regions and human IgG1:k constant regions. Adalimumab (Humira®) is produced by recombinant DNA technology in a mammalian cell expression system and is purified by a process that includes specific viral inactivation and removal steps.
Adalimumab (Humira®) binds specifically to (TNF-a) and blocks its interaction with the p55 and p75 cell surface TNF receptors. Adalimumab (Humira®) also lyses surface TNF expressing cells in vitro in the presence of complement. Adalimumab (Humira®) does not bind or inactivate lymphotoxin (TNF-beta). TNF is a naturally occurring cytokine that is involved in normal inflammatory and immune responses. Elevated levels of TNF are found in the synovial fluid of rheumatoid arthritis, including juvenile idiopathic arthritis, psoriatic arthritis, and ankylosing spondylitis patients and play an important role in both the pathologic inflammation and the joint destruction that are hallmarks of these diseases. Increased levels of TNF are also found in psoriasis (Ps) plaques.
Adalimumab uses
Adalimumab is to treat various autoimmune diseases and works to treat inflammation. Adalimumab is administered to minimize pain and swelling due to various types of arthritis, including rheumatoid arthritis, psoriatic arthritis, juvenile idiopathic arthritis and ankylosing spondylitis. Adalimumab also treats various skin conditions including plaque psoriasis and hidradenitis suppurativa.
Adalimumab immunogenicity
When administered to patients, all therapeutic proteins have the potential to induce an unwanted immune response for example, to stimulate the formation of antidrug antibodies (ADAs). The impact of immune responses can range from no apparent effect to changes in pharmacokinetics, loss of effect and serious adverse events.
According to the manufacturer's product insert, the use of Adalimumab (Humira®) was associated to the development of anti-adalimumab antibodies, even some were reported to be neutralizing in various percentages of patients, during therapy with the drug Humira®. This has the possibility to lead to severe complications. This Assay Genie Antibody to Adalimumab ELISA Kit can be efficiently used for the quantitative determination of Adalimumab anti-drug antibodies in biological samples and is for research use only.
Anti-Adalimumab (Humira®) ADA Quantitative ELISA Kit Contents
| Size | Kit Contents |
1 x 12 x 8 | Microtiter Plate Break apart strips. Microtiter plate with 12 rows each of 8 wells coated with reactant |
7 x 0.3 mL | Adalimumab Standards A-E Standard A: 1000 ng/mL |
0.3mL | Control low and high levels |
1 x 50 mL | Assay Buffer |
1 x 12 mL | Horse radish peroxidase-Conjugated Probe. Red coloured. Ready to use. Contains HRP-probe, stabilizer and preservatives. |
1 x 12 mL | TMB Substrate Solution |
1 x 12 mL | TMB Stop Solution |
1 x 50 mL | Wash Buffer concentrate (20x) |
2 x 1 | Adhesive Foil |
Anti-Adalimumab (Humira®) ADA Quantitative ELISA Protocol
| Steps | Protocol |
1 | Pipette 100µl of Assay Buffer non-exceptionally into each of the wells to be used. |
2 | Pipette 20 µL of each ready-to use Standards, High Level Control, Low Level Control and Diluted Samples into the respective wells of microtiter plate. |
3 | Cover the plate with adhesive foil. Incubate 30 min at room temperature (18- 25°C). |
4 | Remove adhesive foil. Discard incubation solution. Wash plate 3 times each with 300µL of diluted. Wash Buffer. Remove excess solution by tapping the inverted plate on a paper towel. |
5 | Pipette 100 µL of ready-to use HRP-Conjugated Probe into each well. |
6 | Cover the plate with adhesive foil. Incubate 30 min at room temperature (18- 25°C). |
7 | Remove adhesive foil. Discard incubation solution. Wash plate 3 times each with 300 µL of diluted Wash Buffer. Remove excess solution by tapping the inverted plate on a paper towel. |
8 | Pipette 100 µL of TMB Substrate Solution into each well. |
9 | Incubate 10 min (without adhesive foil) at room temperature (18-25°C) in the dark |
10 | Stop the substrate reaction by adding 100 µL of Stop Solution into each well. Briefly mix contents by gently shaking the plate. Colour changes from blue to yellow. |
11 | Measure optical density with a photometer at 450/650 nm within 30 min after pipetting of the Stop Solution. |
Trademarks
Humira® is a registered trademark of AbbVie Biotechnology, Inc.